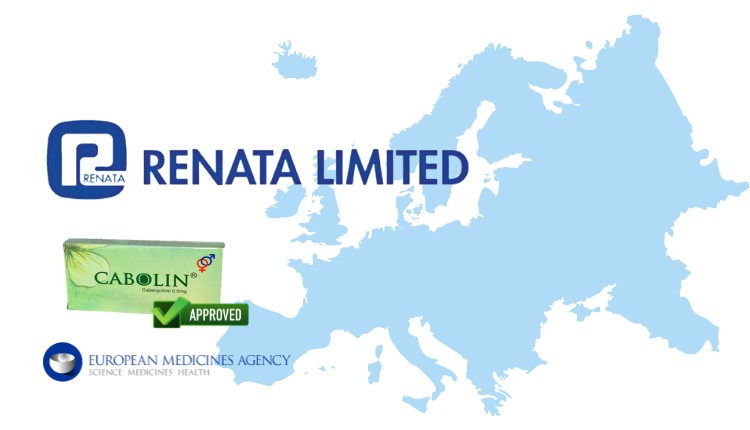
Renata, a leading Bangladeshi pharmaceutical company, has successfully secured approval to export its Cabergoline 0.5mg tablets to several European countries. The drug, used in treating conditions like Parkinson’s disease and hyperprolactinemia, has been cleared through the EU Decentralized Procedure (DCP), enabling its sale across a range of European markets.
The company made the announcement in a filing with the Dhaka Stock Exchange (DSE) on September 2, confirming that the approval grants access to markets including Ireland, France, Portugal, Italy, Denmark, Sweden, the Netherlands, Norway, and Spain. “This important milestone highlights Renata’s commitment to expanding its presence in Europe by developing and delivering high-quality, low-dose, high-potency medicines,” the company stated in its DSE announcement.
The 0.5mg Cabergoline tablets will be produced at Renata’s advanced UK MHRA-approved facility, which follows stringent quality control standards. The facility is designed to meet the specific requirements of the European market, ensuring the medication is produced to the highest quality standards. Renata also confirmed that the product will be distributed across Europe through a network of strategic partnerships, allowing widespread patient access to the treatment.
In addition to the European expansion, Cabergoline is already available in Bangladesh under the brand name Cabolin. This approval further cements Renata’s global standing in the pharmaceutical sector. Notably, last year Renata’s European subsidiary, Renata Pharmaceuticals (Ireland) Limited, received approval from the EU and German regulators to launch Cabergoletten 1mg and 2mg tablets for Parkinson’s treatment in those markets. Renata’s continued success in expanding its product reach reflects its growing influence and capability in the global pharmaceutical industry.
For more updates, be with Markedium.











































Leave a comment